_CERVIX
WIN REMARKABLE OPPORTUNITIES
BY FOLLOWING SPONSORS LINKS
_EPIDEMIOLOGY
In
2007, an estimated 11,150 cases of invasive cervical cancer will be
diagnosed in the U.S., and an estimated 3,670 women will die from this
disease. Globally, cervical cancer is the second most common cause of
cancer death in women, with an estimated 510,000 newly diagnosed
cervical cancer cases and 288,000 deaths. In developing countries,
cervical cancer is often the most common cancer in women. Virtually all
cervical cancers are causally related to infections by HPV.
Approximately 70 percent of cervical cancers are caused by HPV types 16
or 18. About 500,000 precancerous lesions (cervical intraepithelial
neoplasia Grade 2 and 3; CIN2 and CIN3) are diagnosed each year in the
United States, and about 50 to 60 percent are attributable to HPV16 and
HPV18. 4
ETIOLOGY
Human papilloma virus (HPV) infection
For cervical cancer, the most important risk factor is infection with a virus known as HPV (human papilloma virus). HPV is really a group of more than 100 related viruses that can infect cells on the surface of the skin. Some types of HPV cause genital warts. Other types cause cancer of the cervix. The kinds that cause cancer are called "high-risk" HPVs. HPV is passed from one person to another by skin-to-skin contact such as vaginal, anal, or oral sex. But sex isn't the only way to spread HPV from one person to person. All that is needed is for there to be skin-to-skin contact with an area of the body infected with HPV.
Smoking: Women who smoke are about twice as likely to get cervical cancer as those who don't. Smoking puts many chemicals that cause cancer into the lungs. These harmful substances are carried in the bloodstream throughout the body to other organs, too. Tobacco by-products have been found in the cervical mucus of women who smoke.
HIV infection: HIV (human immunodeficiency virus) is the virus that causes AIDS -- it is not the same as HPV. It can also be a risk factor for cancer of the cervix. Having HIV seems to make a woman's immune system less able to fight both HPV and early cancers.
Chlamydia infection: This is a common kind of bacteria that can infect women's sex organs. It is spread during sex. A woman may not know that she is infected at all unless she is tested for chlamydia when she gets her pelvic exam. Some studies suggest that women who have a past or current infection are at greater risk for cancer of the cervix. Long-term infection can cause other serious problems, too.
Diet: What you eat can play a part as well. Diets low in fruits and vegetables are linked to an increased risk of cervical cancer. Also, women who are overweight are at a higher risk.
Birth control pills: Long-term use of birth control pills increases the risk of this cancer. Research suggests that the risk of cervical cancer goes up the longer a woman takes "the pill," but the risk goes back down again after she stops. You should talk to your doctor about the pros and cons of birth control pills in your case.
Having many pregnancies: Woman who have had 3 or more full-term pregnancies have an increased risk of this cancer. No one really knows why this is true.
Young age at the time of first full-term pregnancy: Women who were younger than 17 years when they had their first full-term pregnancy are almost 2 times more likely to get cervical cancer later in life than women who waited to get pregnant until they were 25 years or older.
Low income: Poor women are at greater risk for cancer of the cervix. This may be because they cannot afford good health care, such as regular Pap tests.
DES (diethylstilbestrol): DES is a hormone drug that was used between 1940 and 1971 for some women who were in danger of miscarriages. The daughters of women who took this drug while they were pregnant with them have a slightly higher risk of cancer of the vagina and cervix.
Family history: Cervical cancer may run in some families. If your mother or sister had cervical cancer, your chances of getting the disease are 2 to 3 times higher than if no one in the family had it. This could be because these women are less able to fight off HPV than other women. 11
loading Favorite Sites...
SIGN & SYMPTOMS
IFor pre-cancers and early cancers of the cervix, there are usually no symptoms associated.1 One the disease has progressed and becomes invasive. One of the most coomon earlier symptoms of cervical cancer is postcoital bleeding. This can increase to the point of bleeding between menstrual cycles and/or very heavy menstrual bleeding. There may be a foul-smelling, yellowish discharge with patients that have invasive, necrotic lesions. If one suffers from chronic bleeding due to the disease, they can develop fatigue and other symptoms from anemia. Pain is another symptom that may present itself. This is usually in the pelvis or hypogastrium and can be caused by a necrotic tumor or an associated pelvic inflammatory disease. "Some patients may complain of pain in the lumbosacral area, and in these cases the possibility of paraaortic lymph node involvement with extension into the lumbosacral roots or hydronephrosis should be considered." On occasion, epigastic pain may be reported from metastasis to the high paraaortic lymph nodes. In advanced stages of the disease, urinary and rectal symptoms such as hematuria and rectal bleeding may appear as a result of tumor invasion of the bladder or rectum.2
DIAGNOSTC PROCEDURES
A
general history and physical should be performed on the patient wtih
special attention to the supraclavicular nodal areas, abdomen, liver,
and a gentle but thorough pelvic examination. The "pelvic exam should
include inspection of external genitalia, vagina, and uterine cervix,
rectal examination, and bimanual palpation of the pelvis. Cystoscopy or
rectosigmoidoscopy should be performed in all patients with IIB, III,
and IVA disease or those with earlier stages who have a history of
urinary or lower gastrointestinal tract disturbances."
Women who are 20 years of age and older and those who are younger than 20 that are sexually active are recommended to have a Papanicolaou (Pap) smear yearly for 2 years and then at least 1 every 3 years thereafter until they are 65. This recommendation comes from the American Caner Society. The American College of Obstetricians and Gynecologists strongly recommend continuing to thave them annually. Those who have a higher risk of developing cervical cancer because of early age of first intercourse, having multiple sexual partners, and multiparity should have a pap smear yearly. "If the cytologic smear shows dysplasia or malignant cells, directed biopsies at colposcopy should be carried out immediately. Endocervical curretage must always be performed except in pregnant women. If the biopsy results are negative, the procedure should be repeated and, if necessary, a conization should be performed." Colposcopy evaluates the exocervix and part of the endocervix next to the transition of the squamous and columnar epithelium (T zone). This exam provides a 10 to 15 times magnification of the cervix. This, combined with cytology and biopsy of the abnormal sites, may be useful in detecting the majority of early lesions. "Conization must be performed in spedific situations: when no gross lesion of the cervix is noted and an endocervical tumor is suspected; the entire lesion cannot be seen with the colposcope; diagnosis of microinvasive carcinoma is made on biopsy; discrepancies are found between the cytologic and th histologic appearances of the lesion; or the patient is not reliable for continuous follow-up." Conization consists of removing a large portion of the exo and endocervix. "At least 50% of the endocervical canal should be removed without compromising the internal sphincter."
When there is a gross lesion of the cervix, punch biopsies are considered adequate to confirm the diagnosis of invasive carcinoma. The biopsies should be obtained from tissue at the periphery of the lesion along with some adjoining normal tissue. In addition, biopsies should be taken from all four quadrants of the cervix and any other suspect areas in the cervix or vagina.
Dilation and curettage is recommended at the time of inital evaluation because the possibility of upper extension of the tumor would change the plan of therapy.
"For invasive carcinoma, patients should have the following laboratory studies: complete peripheral blood evaluation, including hemogram, white blood cell count, differential, and platelet count; blood chemistry profile, with particular attention to blood urea nitrogen, creatinine, and uric acid; liver function values; and urinalysis."
Imaging studies:
All patients should have a chest x-ray and an intravenous pyelogram (IVP) for staging purposes. Cat scans with contrast have now taken over the use of IVP's. For patients with stage IIB, III, IVA disease, or those who have symptoms relating to the colon and rectum, a barium enema should be performed.2
Women who are 20 years of age and older and those who are younger than 20 that are sexually active are recommended to have a Papanicolaou (Pap) smear yearly for 2 years and then at least 1 every 3 years thereafter until they are 65. This recommendation comes from the American Caner Society. The American College of Obstetricians and Gynecologists strongly recommend continuing to thave them annually. Those who have a higher risk of developing cervical cancer because of early age of first intercourse, having multiple sexual partners, and multiparity should have a pap smear yearly. "If the cytologic smear shows dysplasia or malignant cells, directed biopsies at colposcopy should be carried out immediately. Endocervical curretage must always be performed except in pregnant women. If the biopsy results are negative, the procedure should be repeated and, if necessary, a conization should be performed." Colposcopy evaluates the exocervix and part of the endocervix next to the transition of the squamous and columnar epithelium (T zone). This exam provides a 10 to 15 times magnification of the cervix. This, combined with cytology and biopsy of the abnormal sites, may be useful in detecting the majority of early lesions. "Conization must be performed in spedific situations: when no gross lesion of the cervix is noted and an endocervical tumor is suspected; the entire lesion cannot be seen with the colposcope; diagnosis of microinvasive carcinoma is made on biopsy; discrepancies are found between the cytologic and th histologic appearances of the lesion; or the patient is not reliable for continuous follow-up." Conization consists of removing a large portion of the exo and endocervix. "At least 50% of the endocervical canal should be removed without compromising the internal sphincter."
When there is a gross lesion of the cervix, punch biopsies are considered adequate to confirm the diagnosis of invasive carcinoma. The biopsies should be obtained from tissue at the periphery of the lesion along with some adjoining normal tissue. In addition, biopsies should be taken from all four quadrants of the cervix and any other suspect areas in the cervix or vagina.
Dilation and curettage is recommended at the time of inital evaluation because the possibility of upper extension of the tumor would change the plan of therapy.
"For invasive carcinoma, patients should have the following laboratory studies: complete peripheral blood evaluation, including hemogram, white blood cell count, differential, and platelet count; blood chemistry profile, with particular attention to blood urea nitrogen, creatinine, and uric acid; liver function values; and urinalysis."
Imaging studies:
All patients should have a chest x-ray and an intravenous pyelogram (IVP) for staging purposes. Cat scans with contrast have now taken over the use of IVP's. For patients with stage IIB, III, IVA disease, or those who have symptoms relating to the colon and rectum, a barium enema should be performed.2
HISTOLOGY
More
than 90% of cervical tumors are squamous cell carcinoma. Adenocarcinoma
accounts for approximately 7% to 10%, and 1% to 2% are clear cell,
mesonephric type.2
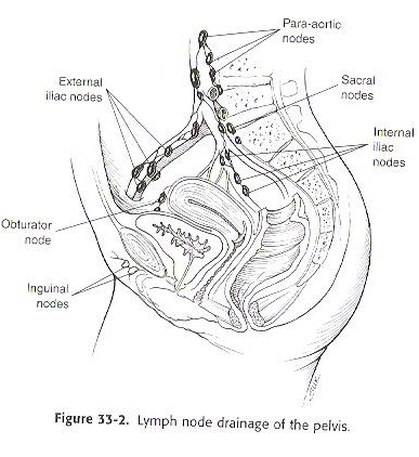
LYMPH NODE DRAINAGE
The
lymph node drainage of the pelvis includes the inguinal lymph nodes (
external and internal), pelvic nodes ( the internal iliac chain , that
originates at with the obturator node , and external iliac chain), and
periaortic nodes.5 See figure 1.1.
METASTATIC SPREAD
Carcinoma
of the uterine cervix may spread to the vaginal fornices or
paracervical and parametrial tissues. Untreated carcinomas can cause
direct invasion of the bladder , rectum or both. The most common
metastatic sites include; the lungs, paraaortic nodes, mediastinal and
superclavicular lymph nodes, bones, and liver. 3
GRADING
Once the biopsy is taken , the grading is done on the sample. The grading is determined by how much the cells look like normal. The grading is a measurement of aggressiveness of the disease. 6
Grade 1 ( low grade) is well differentiated. The cancer cells appear similar to normal cells and are slow growing.
Grade 2 (moderate grade) These cells look slightly different and are slightly faster growing.
Grade 3 ( High grade) cells are poorly differentiated. The cells look abnormal and grow faster. These cells tend to spread faster.
Grade 1 ( low grade) is well differentiated. The cancer cells appear similar to normal cells and are slow growing.
Grade 2 (moderate grade) These cells look slightly different and are slightly faster growing.
Grade 3 ( High grade) cells are poorly differentiated. The cells look abnormal and grow faster. These cells tend to spread faster.
STAGING
It is imperative that the gynecologic and radiation oncologist jointly stage the tumor in every patient, with bimanual pelvic and rectal examination under general anesthesia.
Key:
AJCC - American Joint Committee on Cancer
(FIGO) - International Federation of Gynecologists and Oncologist
Primary tumor (T)
TX: Primary tumor cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1 (I): Cervical carcinoma confined to uterus (extension to the corpus should be disregarded)
T1a (IA): Preclinical invasive carcinoma, diagnosed by microscopy onlya
T1a1 (IA1): Measured stromal invasion less than or equal to 3 mm in depth and less than or equal to 7 mm in horizontal spread
T1a2 (IA2): Measured stromal invasion greater than 3 mm and less than 5 mm with a horizontal spread of less than or equal to 7 mm
T1b (IB): Clinically visible lesion confined to the cervix or microscopic lesion greater than T1a/IA2
T1b1 (IB1): Clinically visible lesion less than or equal to 4 cm in greatest dimension
T1b2 (IB2): Clinically visible lesion greater than 4 cm in greatest dimesion
T2 (II): Cervical carcinoma invades beyond uterus but not to the pelvic wall or to the lower third of vagina
T2a (IIA): Tumor without parametrial invasion
T2b (IIB): Tumor with parametrial invasion
T3 (III): Tumor extends to the pelvic wall, and/or involves the lower third of the vagina, and/or causes hydronephrosis or nonfunctioning kidney
T3a (IIIA): Tumor involves lower third of vagina, no extension to pelvic wall
T3b (IIIB): Tumor extends to pelvic wall and/or causes hydronephrosis or nonfunctioning kidney
T4b (IVA): Tumor invades mucosa of the bladder or rectum, and/or extends beyond the true pelvis
M1 (IVB): Distant metastasis
Regional lymph nodes (N)
-Regional lymph nodes include paracervical, parametrial, hypogastric (obturator), common, internal and external iliac, presacral, and sacral
NX: Regional lymph nodes cannot be assessed
N0: No regional lymph node metastasis
N1: Regional lymph node metastasis
Distant metastasis (M)
MX: Presence of distant metastasis cannot be assessed
M0: No distant metastasis
M1: Distant metastasis 3
a All macroscopically visible lesions-even with superficial invasion-are T1b/IB. Stromal invasion with a maximal depth of 5 mm measured from the base of the epithelium and a horizontal spread of 7 mm or less. Vascular space involvement, venous or lymphatic, does not affect classification.
bBullous edema is not sufficient to classify a tumor as T4.
Key:
AJCC - American Joint Committee on Cancer
(FIGO) - International Federation of Gynecologists and Oncologist
Primary tumor (T)
TX: Primary tumor cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1 (I): Cervical carcinoma confined to uterus (extension to the corpus should be disregarded)
T1a (IA): Preclinical invasive carcinoma, diagnosed by microscopy onlya
T1a1 (IA1): Measured stromal invasion less than or equal to 3 mm in depth and less than or equal to 7 mm in horizontal spread
T1a2 (IA2): Measured stromal invasion greater than 3 mm and less than 5 mm with a horizontal spread of less than or equal to 7 mm
T1b (IB): Clinically visible lesion confined to the cervix or microscopic lesion greater than T1a/IA2
T1b1 (IB1): Clinically visible lesion less than or equal to 4 cm in greatest dimension
T1b2 (IB2): Clinically visible lesion greater than 4 cm in greatest dimesion
T2 (II): Cervical carcinoma invades beyond uterus but not to the pelvic wall or to the lower third of vagina
T2a (IIA): Tumor without parametrial invasion
T2b (IIB): Tumor with parametrial invasion
T3 (III): Tumor extends to the pelvic wall, and/or involves the lower third of the vagina, and/or causes hydronephrosis or nonfunctioning kidney
T3a (IIIA): Tumor involves lower third of vagina, no extension to pelvic wall
T3b (IIIB): Tumor extends to pelvic wall and/or causes hydronephrosis or nonfunctioning kidney
T4b (IVA): Tumor invades mucosa of the bladder or rectum, and/or extends beyond the true pelvis
M1 (IVB): Distant metastasis
Regional lymph nodes (N)
-Regional lymph nodes include paracervical, parametrial, hypogastric (obturator), common, internal and external iliac, presacral, and sacral
NX: Regional lymph nodes cannot be assessed
N0: No regional lymph node metastasis
N1: Regional lymph node metastasis
Distant metastasis (M)
MX: Presence of distant metastasis cannot be assessed
M0: No distant metastasis
M1: Distant metastasis 3
a All macroscopically visible lesions-even with superficial invasion-are T1b/IB. Stromal invasion with a maximal depth of 5 mm measured from the base of the epithelium and a horizontal spread of 7 mm or less. Vascular space involvement, venous or lymphatic, does not affect classification.
bBullous edema is not sufficient to classify a tumor as T4.
RADIATION SIDE EFFECTS
Acute Side Effects:
Late Side Effects:
- Acute gastrointestinal side effects of pelvic radiation include diarrhea, abdominal cramping, rectal discomfort, and occasionally, rectal bleeding, which may be caused by transient enteroproctitis.
- Genitourinary symptoms, secondary to cystourethritis, are dysuria, frequency, and nocturia. The urine is usually clear, although there may be microscopic or even gross hematuria.
- Urinary tract infections may occur.
- Erythema and dry or moist desquamation may develop in the perineum or intergluteal fold.
- Acute radiation vaginitis, superficial ulceration of the vagina, and vagianl stenosis can also occur.
Late Side Effects:
- Late radiation effects are closely related to total doses given to the pelvic organs.
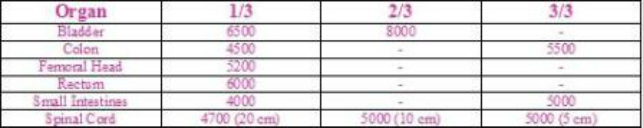
- Maximum radiation doses tolerated by limited volumes are 75 Gy for the rectum and 80 Gy for the urinary bladder when LDR brachytherapy is used.
- Retrovaginal or vesicovaginal fistula and proctitis or cystitis can occur but in small percentages.
- Injury to the gastrointestinal tract usually appears within 2 years of radiation therapy; complications of the urinary tract more frequently are seen 3- 4 years after treatment.
- Vaginal stenosis is associated with dyspareunia in 31% of women treated for carcinoma of the cervix.
- Anal incontinence is observed occasionally. 3
PROGNOSIS
Survival rates and
local control decrease as the stage and bulk of the disease increase.
Ureter invasion has been associated with a reduction in 5 year survival
rates from 92% to 54%. Bulky or barrel-shaped cervical cancer is
associated with a 15% to 20% increase in distant metastses and a
decrease in local control. For stages IB and IIA, the incolvement of
lymph nodes results in approximately a 50% reduction in survival rares. 12
loading Favorite Sites...
TREATMENT
“Treatment should involve close collaboration between the gynecologic oncologist and the radiation oncologist, with an integrated team approach vigorously pursued.” 3
Carcinoma In Situ:
- Is usually treated with a total abdominal hysterectomy, with or without small vaginal cuff. The removal of ovaries depends on the patient’s desire for children and the overall condition of the ovaries.
-Carcinoma In Situ may be treated with therapeutic conization, laser and cryotherapy to preserve fertility.
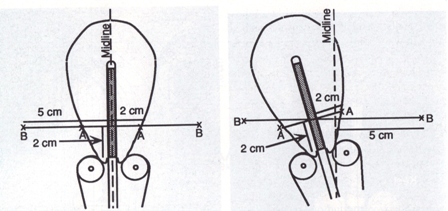
-Tandem and ovoids (intracavitary irradiation) is useful for carcinoma in situ. A dose of 45 to 50 Gy is prescribed to point A. 3
Stage IA:
-Is usually treated with a total abdominal hysterectomy or modified radical hysterectomy. Intracavitary irradiation is an option for stage IA carcinoma of the cervix. Approximately, 56 to 70 Gy to point A. Lymph node dissection and/or pelvic external irradiation is not required with the low risk of lymph node metastases.3
Stage IB and IIA:
-Definitive radiation and radical surgery (similar tumor control and survival) remains highly controversial and depends on patient and lesion specific factors.
-Preoperative doses of 20 to 45 Gy to the whole pelvis, parametrial boost to 50 Gy and intracavitary insertions with 50 Gy to point A (70 Gy total) can be used. An extrafascial hysterectomy follows approximately 4 to 6 weeks after irradiation.
-Irradiation alone (85 to 90 Gy to point A) can be used with tumors larger than 4 cm.
-Irradiation, chemotherapy or both is valuable as adjuvant treatment of squamous cell carcinoma of the cervix after radical hysterectomy.3
Stage IIB, III, IVA:
-“Stage IIB and III are treated with irradiation alone.”
-“Stage IVA (bladder and rectal invasion) can be treated either with pelvic exenteration, or with high doses of external irradiation to the whole pelvis, intracavitary insertion, and additional parametrial irradiation.”
-The use of chemotherapy (hydroxyurea, cisplatin, and 5-fluorouracil combined with mitocycin- C) and irradiation can cause a radiosensitizing effect when used concomitantly. 3
Small Cell Carcinoma:
-Treated similarly to other stages.
-The chemotherapy drugs used the most include cyclophosphamide, doxorubicin (adriamycin) and vincristine sulfate. The use of chemotherapy may allow up to a 10% decrease in the radiation dose.3
Postoperative Irradiation after Radical Hysterectomy:
-Indications for postoperative radiation include high-risk prognostic factors, positive pelvic lymph nodes positive or close margins, deep stromal invasion, or vascular/lymphatic permeation.
-Caution should be taken when designing treatment fields because of changes in and vascular supply.
-“Surgical extirpation of the uterus, the bladder and rectosigmoid may be closer to the radioactive source (intracavitary insertion) than in a patient with an intact uterus.”
-Metastatic lymph nodes receive doses of 50 Gy to the whole pelvis using a four field technique. Positive paraaortics or common iliac nodes receive doses of 50Gy to the periaortic region.
-Deep stromal invasion or close positive margins recieve20 Gy to the whole pelvis, 30 Gy to the parametria (small midline block) and LDR of 65 Gy to the vaginal mucosa with two colpostats (or HDR of three insertions of 6 to 7 Gy at 0.5 cm).3
Residual Tumor:
-20 Gy to the whole pelvis is delivered and 30 Gy to the parametrium with an LDR to the vaginal vault to 60 Gy mucosal dose.
-Interstital implants may also be used to increase the dose to the target volume. 3
Radiation Therapy Techniques
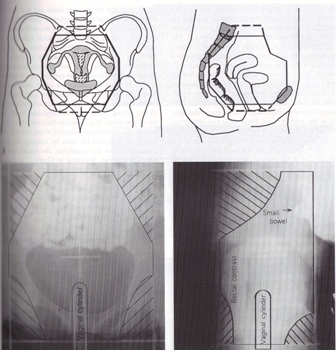
Anterioposterior / Posteroanterior Treatment Borders:
-Superior: should be at L4-5 interspace and include all of the external iliac and hypogastric lymph nodes
(margin is extended to L3 if common iliac nodes are to be treated).
-Inferior: inferior border of obturator foramen (if no vaginal extension)
Include entire organ down to introitus (vaginal extension)
-Width: Level at the obturator fossae is approximately 12.3 cm
Level of the inguinal rings (around the femoral arteries) is approximately 14.6 cm.
“A 2 cm margin lateral to the bony pelvis is adequate.”
-Posterior: posterior to the rectum and extend to the sacral hollow.3
Carcinoma In Situ:
- Is usually treated with a total abdominal hysterectomy, with or without small vaginal cuff. The removal of ovaries depends on the patient’s desire for children and the overall condition of the ovaries.
-Carcinoma In Situ may be treated with therapeutic conization, laser and cryotherapy to preserve fertility.
-Tandem and ovoids (intracavitary irradiation) is useful for carcinoma in situ. A dose of 45 to 50 Gy is prescribed to point A. 3
Stage IA:
-Is usually treated with a total abdominal hysterectomy or modified radical hysterectomy. Intracavitary irradiation is an option for stage IA carcinoma of the cervix. Approximately, 56 to 70 Gy to point A. Lymph node dissection and/or pelvic external irradiation is not required with the low risk of lymph node metastases.3
Stage IB and IIA:
-Definitive radiation and radical surgery (similar tumor control and survival) remains highly controversial and depends on patient and lesion specific factors.
-Preoperative doses of 20 to 45 Gy to the whole pelvis, parametrial boost to 50 Gy and intracavitary insertions with 50 Gy to point A (70 Gy total) can be used. An extrafascial hysterectomy follows approximately 4 to 6 weeks after irradiation.
-Irradiation alone (85 to 90 Gy to point A) can be used with tumors larger than 4 cm.
-Irradiation, chemotherapy or both is valuable as adjuvant treatment of squamous cell carcinoma of the cervix after radical hysterectomy.3
Stage IIB, III, IVA:
-“Stage IIB and III are treated with irradiation alone.”
-“Stage IVA (bladder and rectal invasion) can be treated either with pelvic exenteration, or with high doses of external irradiation to the whole pelvis, intracavitary insertion, and additional parametrial irradiation.”
-The use of chemotherapy (hydroxyurea, cisplatin, and 5-fluorouracil combined with mitocycin- C) and irradiation can cause a radiosensitizing effect when used concomitantly. 3
Small Cell Carcinoma:
-Treated similarly to other stages.
-The chemotherapy drugs used the most include cyclophosphamide, doxorubicin (adriamycin) and vincristine sulfate. The use of chemotherapy may allow up to a 10% decrease in the radiation dose.3
Postoperative Irradiation after Radical Hysterectomy:
-Indications for postoperative radiation include high-risk prognostic factors, positive pelvic lymph nodes positive or close margins, deep stromal invasion, or vascular/lymphatic permeation.
-Caution should be taken when designing treatment fields because of changes in and vascular supply.
-“Surgical extirpation of the uterus, the bladder and rectosigmoid may be closer to the radioactive source (intracavitary insertion) than in a patient with an intact uterus.”
-Metastatic lymph nodes receive doses of 50 Gy to the whole pelvis using a four field technique. Positive paraaortics or common iliac nodes receive doses of 50Gy to the periaortic region.
-Deep stromal invasion or close positive margins recieve20 Gy to the whole pelvis, 30 Gy to the parametria (small midline block) and LDR of 65 Gy to the vaginal mucosa with two colpostats (or HDR of three insertions of 6 to 7 Gy at 0.5 cm).3
Residual Tumor:
-20 Gy to the whole pelvis is delivered and 30 Gy to the parametrium with an LDR to the vaginal vault to 60 Gy mucosal dose.
-Interstital implants may also be used to increase the dose to the target volume. 3
Radiation Therapy Techniques
Anterioposterior / Posteroanterior Treatment Borders:
-Superior: should be at L4-5 interspace and include all of the external iliac and hypogastric lymph nodes
(margin is extended to L3 if common iliac nodes are to be treated).
-Inferior: inferior border of obturator foramen (if no vaginal extension)
Include entire organ down to introitus (vaginal extension)
-Width: Level at the obturator fossae is approximately 12.3 cm
Level of the inguinal rings (around the femoral arteries) is approximately 14.6 cm.
“A 2 cm margin lateral to the bony pelvis is adequate.”
-Posterior: posterior to the rectum and extend to the sacral hollow.3
PLANING PICTURES
Lateral Treatment Field Borders:
-Anterior: cortex of pubic symphysis (should cover the external iliac nodes)
-Posterior: covers at least 50% of the rectum (stage IB) or extended to sacral hollow (more advanced)3
-Anterior: cortex of pubic symphysis (should cover the external iliac nodes)
-Posterior: covers at least 50% of the rectum (stage IB) or extended to sacral hollow (more advanced)3
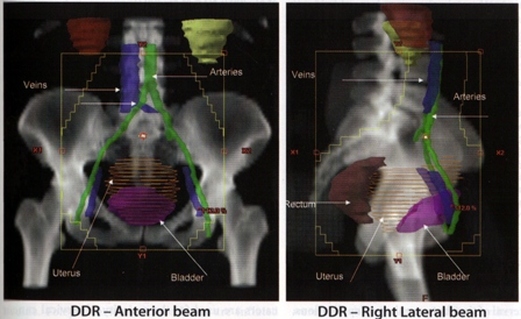
Paraaortics:
-Paraaortics are irradiated with an extended field ( includes paraaortic nodes and the pelvis) or two separate fields requiring a gap calculation ( helps to avoid overlap to small bowel).
-Upper margin: T12-L1 interspace
-Width: determined by CT scan, lymphangiogram or an IVP showing the ureters.3
-Paraaortics are irradiated with an extended field ( includes paraaortic nodes and the pelvis) or two separate fields requiring a gap calculation ( helps to avoid overlap to small bowel).
-Upper margin: T12-L1 interspace
-Width: determined by CT scan, lymphangiogram or an IVP showing the ureters.3
Brachytherapy:
-137Cs (Cesium) is the most popular radioactive isotope used in LDR treatment of cervical carcinoma.
-192 Ir (Iridium) is used for HDR treatment.
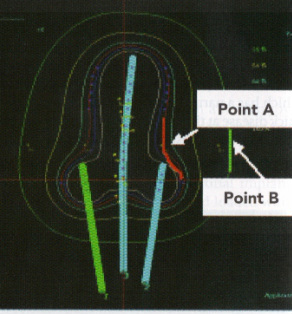
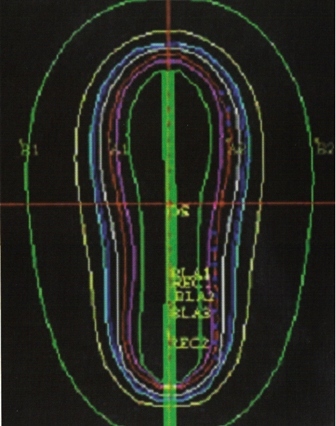
-Intracavitary techniques use intrauterine tandems and vaginal colpostats. Vaginal cylinders can also be used. Radiographs are taken to ensure proper device placement. “An intrauterine tandem with three or four 137Cs sources is inserted into the uterus, and two colpostats are placed in the vaginal vault and packed with iodoformed gauze, to deliver 0.6 to 0.8 Gy per hour (LDR).” 3
-137Cs (Cesium) is the most popular radioactive isotope used in LDR treatment of cervical carcinoma.
-192 Ir (Iridium) is used for HDR treatment.
-Intracavitary techniques use intrauterine tandems and vaginal colpostats. Vaginal cylinders can also be used. Radiographs are taken to ensure proper device placement. “An intrauterine tandem with three or four 137Cs sources is inserted into the uterus, and two colpostats are placed in the vaginal vault and packed with iodoformed gauze, to deliver 0.6 to 0.8 Gy per hour (LDR).” 3
-“Computer- generated isodose curves provide the best means of determining the doses to point A, point B, bladder, and rectum.” 3
-In general the first intracavitary insertion happens following the delivery of between 10 to 40Gy of external whole pelvis irradiation. The second ( usually 1-4 sessions)sessions are scheduled with one to two weeks between each session, following the initial insertion. 3
- “The HDR dose to point A is approximately 0.5 to 0.6 of the LDR dose. A frequently used dose per fraction is 5.0 to 7.5 Gy, 4 to8 fractions, depending upon whole pelvis dose.” 3
Chemotherapy
" Chemotherapy may be used for the treatment of micrometastasis (adjuvant therapy) or in patients with extensive tumors (definitive) or for disseminated disease (palliative)." 3
-In general the first intracavitary insertion happens following the delivery of between 10 to 40Gy of external whole pelvis irradiation. The second ( usually 1-4 sessions)sessions are scheduled with one to two weeks between each session, following the initial insertion. 3
- “The HDR dose to point A is approximately 0.5 to 0.6 of the LDR dose. A frequently used dose per fraction is 5.0 to 7.5 Gy, 4 to8 fractions, depending upon whole pelvis dose.” 3
Chemotherapy
" Chemotherapy may be used for the treatment of micrometastasis (adjuvant therapy) or in patients with extensive tumors (definitive) or for disseminated disease (palliative)." 3
TD5/5
Table provided by Wikibooks.10
"In LDR therapy, it is common to limit the the total bladder and rectal doses 75 to 70 Gy, unless it would compromise tumor control. In HDR, the total doses will be lower, but it is not clear how much lower they are." 9
"In LDR therapy, it is common to limit the the total bladder and rectal doses 75 to 70 Gy, unless it would compromise tumor control. In HDR, the total doses will be lower, but it is not clear how much lower they are." 9